What we do
We develop and commercialize innovative medicines in the anti-infectives therapy area.

We have a proven track record of bringing drugs from research through clinical development all the way to the market.
Our portfolio
Disease areas
We develop and commercialize innovative medicines in the anti-infectives therapy area.
Our products and pipeline
Anti-infectives, specifically antifungals and antibiotics, is the key focus of Basilea’s strategy. We have successfully brought two anti-infective drugs to the market: the antifungal Cresemba (isavuconazole) and the antibiotic Zevtera (ceftobiprole).
Cresemba
Isavuconazole, marketed under its trade name Cresemba, is an intravenous and oral antifungal drug for the treatment of the two most frequent invasive mold infections: invasive aspergillosis and mucormycosis.
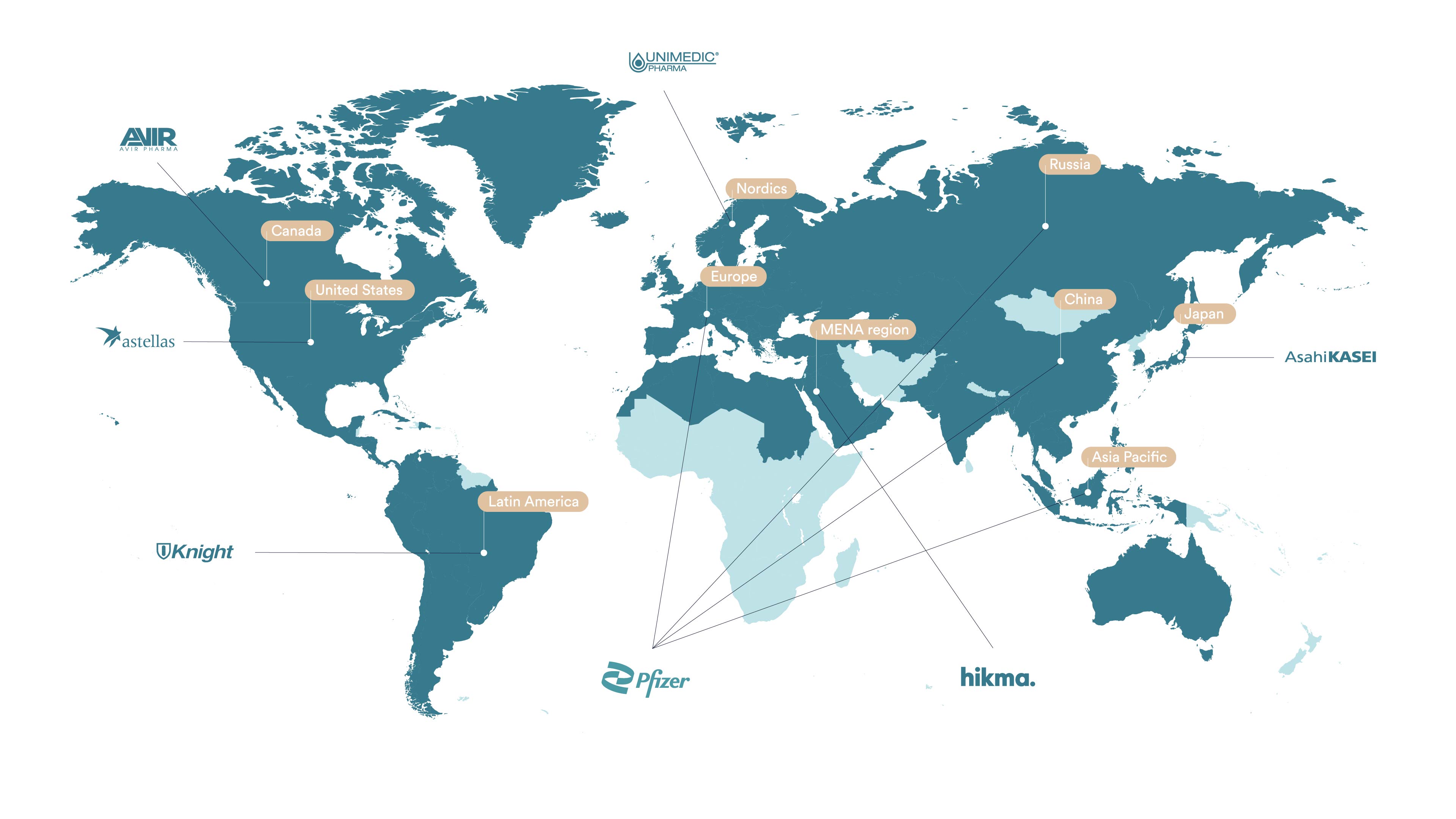
Basilea has established several license and distribution agreements for isavuconazole covering well over 100 countries. The partners include Astellas Pharma Inc. for the U.S. and Pfizer for most of Europe (excluding the Nordic countries, where Unimedic is our distribution partner), Russia, Turkey, Israel, China and further countries in Asia Pacific. Moreover, we have strong regional partners for Latin America (Knight), Japan (Asahi Kasei Therapeutics), the MENA region, i.e. Middle East and North Africa, (Hikma) and Canada (Avir).
Isavuconazol is approved in more than 70 countries world-wide, including the EU member states, the US, China and Japan. The registration status and approved indications may vary from country to country. It has orphan drug designation in the US, Europe and Australia for its approved indications. In addition, Cresemba has been approved for the use in children in the US and the EU.
Basilea participates in the commercial success of Cresemba sales through royalties, regulatory and sales milestone payments by Basilea’s commercialization partners, and by selling Cresemba to the distribution partners at a transfer price.

Global partnerships Cresemba
Fosmanogepix
Fosmanogepix is a clinical-stage broad-spectrum antifungal candidate with a novel mechanism of action. Basilea has acquired this first-in-class candidate in November 2023. Fosmanogepix is available in intravenous and oral formulations and has been evaluated for efficacy and safety in clinical phase 2 studies for the treatment of patients with Candidemia, including Candida auris, and invasive mold infections. In September 2024 Basilea initiated a phase 3 study with fosmanogepix in candidemia and invasive candidiasis; and in July 2025 a second phase 3 study in invasive mold infections.
This project is funded in part with federal funds from the US Department of Health and Human Services (HHS); Administration of Strategic Preparedness and Response (ASPR); Biomedical Advanced Research and Development Authority (BARDA), under OTA number: 75A50124C00033.
BAL2062
BAL2062 (formerly GR-2397) is a clinical-stage antifungal compound with a novel mechanism of action, targeting invasive mold infections. Basilea has acquired this first-in-class antifungal in October 2023. It is derived from a natural product, and has demonstrated fungicidal activity against clinically important molds such as Aspergillus species, including azole-resistant strains. Safety and tolerability have been demonstrated in a previously completed phase 1 study with single and multiple ascending intravenous doses. The drug candidate has Qualified Infectious Disease Product (QIDP), Orphan Drug and Fast Track designation from the US Food & Drug Administration (FDA) for invasive aspergillosis.
This project is funded in part with federal funds from the US Department of Health and Human Services (HHS); Administration of Strategic Preparedness and Response (ASPR); Biomedical Advanced Research and Development Authority (BARDA), under OTA number: 75A50124C00033.
Zevtera
Ceftobiprole, marketed in most countries under the trade name Zevtera, is an intravenous antibiotic currently approved for the treatment of pneumonia, especially the pneumonia acquired in hospitals.
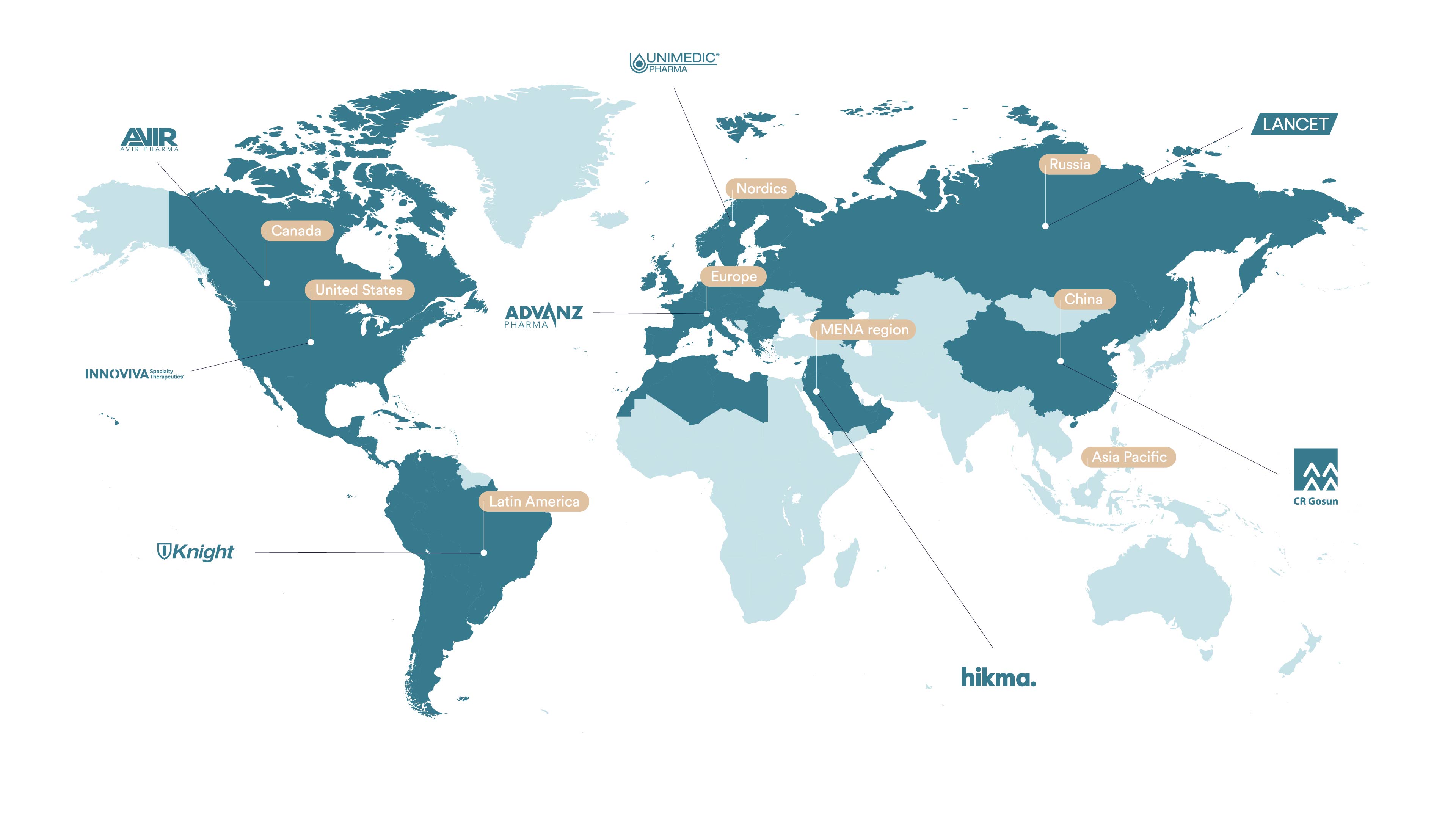
We have established license and distribution agreements for ceftobiprole with several partners covering more than 80 countries. This includes Innoviva Specialty Therapeutics for the United States, Advanz for Europe (excluding the Nordic countries, where Unimedic is our distribution partner), as well as strong regional partners for Latin America (Knight), the MENA region (Hikma), China (CR Gosun), Canada (Avir) and Eurasian countries (JSC Lancet).
As with Cresemba, we participate in the commercialization of Zevtera through royalties, regulatory and sales milestone payments, and by selling the drug to our distributors.
In April 2024, the US Food and Drug Administration (FDA) approved our New Drug Application (NDA) for ceftobiprole for the treatment of Staphylococcus aureus bacteremia (SAB), including right-sided infective endocarditis, acute bacterial skin and skin structure infections (ABSSSI) and also for community-acquired bacterial pneumonia (CABP).
In May 2025, Zevtera (ceftobiprole medocaril for injection) has become commercially available in the United States through our partner Innoviva Specialty Therapeutics.
The ceftobiprole phase 3 program was funded in part with federal funds from the US Department of Health and Human Services (HHS); Administration for Strategic Preparedness and Response (ASPR); Biomedical Advanced Research and Development Authority (BARDA), under contract number HHSO100201600002C. Basilea has been awarded approximately USD 112 million, or approximately 75 percent of the costs related to the SAB and ABSSSI phase 3 studies, regulatory activities and non-clinical work.
Global partnerships Zevtera
Ceftibuten-ledaborbactam etzadroxil
Ledaborbactam etzadroxil is the orally bioavailable prodrug of ledaborbactam, a novel broad-spectrum boronic acid beta-lactamase inhibitor, which is being developed in combination with ceftibuten, an oral beta-lactam antibiotic of the cephalosporin class, which is approved in the US for the treatment of upper and lower respiratory tract infections and for urinary tract infections outside the US.
Basilea acquired the global rights to the beta-lactam/beta-lactamase inhibitor (BL/BLI) combination in August 2025 and expects initiating a registrational phase 3 program in cUTI in early 2027.
In vitro and in vivo studies demonstrated that ledaborbactam etzadroxil restores the activity of ceftibuten against strains of Enterobacterales expressing Ambler class A extended spectrum beta-lactamases (ESBLs), class C cephalosporinases, and class A and D carbapenemases (KPC and OXA-48, respectively) as well as multidrug-resistant (MDR) Enterobacterales. Ceftibuten-ledaborbactam etzadroxil has been granted Qualified Infectious Disease Product (QIDP) and Fast Track designations by the US Food and Drug Administration (FDA) for cUTI and uncomplicated urinary tract infections. Ceftibuten-ledaborbactam etzadroxil is an investigational drug and is not yet approved in any country for commercial use.
BAL2420 (LptA inhibitor)
BAL2420 belongs to one of the very few novel classes of antibiotics in development. It is targeting LptA, which is part of the lipopolysaccharide transport bridge, an essential structure in Gram-negative bacteria. LptA inhibitors have shown potent and rapid bactericidal activity in vitro and in vivo against Gram-negative bacteria of the Enterobacteriaceae family, such as E. coli and K. pneumoniae, including strains resistant to beta-lactams and colistin, an antibiotic regarded as last-resort therapy. Enterobacteriaceae have been highlighted by the World Health Organization as priority pathogens, against which new antibiotics are urgently needed.
In March 2026, a first-in-human phase 1 study (clinicaltrials.gov identifier NCT07500181) was started, evaluating the safety, tolerability and pharmacokinetics of BAL2024 in healthy adult volunteers. The single-center, randomized, dose-escalation, double-blind and placebo-controlled study assesses intravenous administration of BAL2420 using a single‑ and multiple‑ascending dose study design. Data from this study will support the further clinical development of BAL2420 as a potential treatment option for serious infections caused by Gram‑negative bacteria, including multidrug-resistant bacteria.
This project has been awarded a grant from CARB-X (Combating Antibiotic-Resistant Bacteria Biopharmaceutical Accelerator), a global non-profit partnership dedicated to supporting the early development of antibacterial products. CARB-X’s funding for this project is provided by federal funds from the US Department of Health and Human Services; Administration for Strategic Preparedness and Response; Biomedical Advanced Research and Development Authority; under agreement number: 75A50122C00028, and by awards from Wellcome (WT224842) and Germany’s Federal Ministry of Research, Technology and Space (BMFTR).